
The sample is then treated with dilute HCl, washed with deionized water and dried at 105✬. The sample is then washed with 0.1 M NaOH to remove possible contamination by humic acids. The sample is placed 1N HCl and heated to 80✬ for 1 hour, centrifuged and decanted. For small or poorly preserved samples, the alkali treatment may be shortened or omitted completely, or humic acids may precipitated out of alkali solution for radiocarbon dating. This involves three steps: (1) an acid treatment to remove secondary carbonates and acid-soluble compounds (2) an alkali treatment to separate out humic acids and (3) a second acid treatment to remove atmospheric CO 2. The AAA method is used to pretreat a wide variety of sample types including plant material, charcoal, wood, soils, sediment, peat, and plant-based textiles. However, the implementation of these techniques may vary depending on the size and condition of the sample. Certain chemical pretreatment techniques are considered routine for specific sample types or contaminants, and are described below. The goal of chemical pretreatment is to remove contaminants that are chemically soluble. In some cases, samples are sieved to select an appropriate size fraction, or gently crushed to reduce the size of the particles. Depending on the sample type, surface dirt may be removed by washing in an ultrasonic bath or by physically removing the outermost layer of the sample using a rotary tool or scalpel. Some common contaminants include intrusive rootlets, which are manually separated from the sample using forceps, and surface dirt. This may involve cleaning to remove obvious contaminants and/or reduction of particle size of the sample.Ĭleaning involves the physical, rather than chemical, removal of obviously intrusive materials. In many cases further physical pretreatment is required. Communication between the radiocarbon researcher and the sample collector is integral to this process.Īll samples are physically examined to evaluate the composition and preservation of the sample, and to determine the appropriate pretreatment plan. Selecting the appropriate pretreatment plan depends on the unique attributes of the sample itself, such as the sample type, potential contaminants, the burial context, and the size and preservation of the sample. The goal of sample pretreatment is to isolate the carbon fraction required for radiocarbon dating and to remove carbon fractions that are altered or contaminated. Contaminants are carbon-containing materials that are not indigenous to the original organic material being dated. Hope this helps, I know I've repeated things that you know but in case there's anything you missed.In general, it should be assumed that all samples are affected by some form of alteration or contamination. Their methods of finding the ratio is extremely accurate, so unless $\Delta t$ is under 20 years, I suspect radiocarbon dating will work. To continue further, carbon daters find the ratio between carbon-14 and carbon-12 (which does not decay) to find $N$ and $N_0$. 
For your situation, all that is known is the half-life of carbon-14. 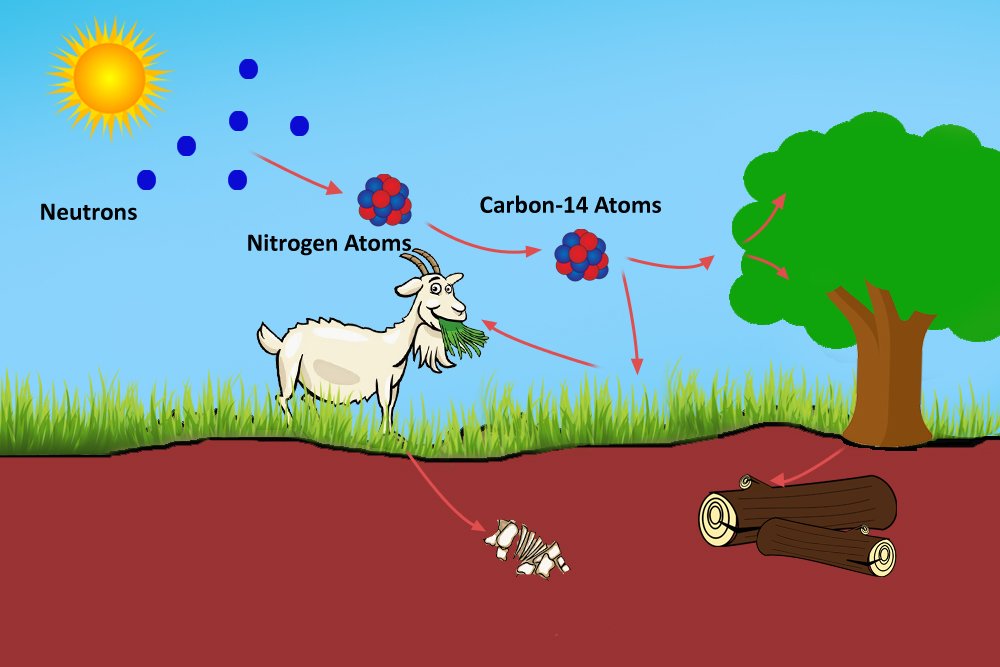
it is very reliable for your $\approx$ 200 year old wood). This is enough information to calculate the age of any organic, or living material, up to an age of around 40 000 to 50 000 years (i.e. Where $\Delta t$ represents the time elapsed (or change in time), and $\lambda$ represents the half-life (in years). Where $N$ represents the mass of radioisotope remaining, $N_0$ represents the mass of radioisotope at the beginning, and $n$ is the amount of half-lives that have passed. The basic formulae that are required for radioisotopes and nuclear half-life:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
